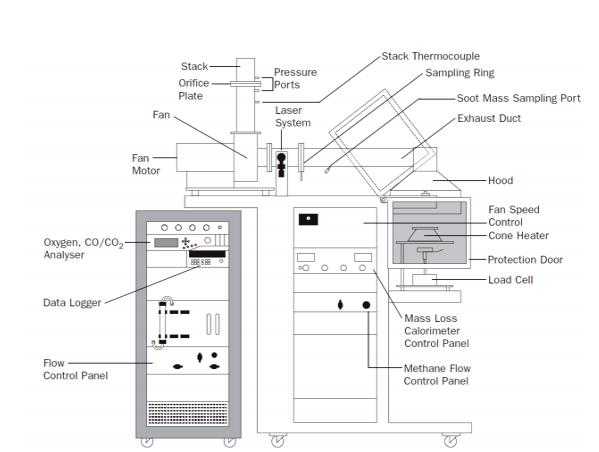
The cone calorimeter consists of numerous essential parts and devices. Together these parts measure, log, set and adjust several parameters like temperatures, mass, gas flow and concentration.
The specimen is placed on a metal sample holder, which is placed on the load cell. The load cell records the weight of the sample during the experiment. There are two types of sample holders. Depending on the specimen, the sample holder can have open or closed edges. If the specimen is of intumescent type (i.e. the specimen swells and forms a protective char layer), a wired grid can be fitted to keep the specimen in place when swelling occurs. The wired grid is always used when testing in horizontal position and with the edged frame sample holder.
A spark igniter is situated right above the sample surface below the cone heater. This ignites the flammable gases leaving the sample when the sample is heated. When the whole sample area is burning, the igniter is turned off and moved to the side. Prior to the experiment during calibration, a water cooled heat flux measuring device is placed at the sample surface level. The cone heater is then set to the temperature that gives the wanted heat flux at the surface of the specimen. Normally, the cone is mounted horizontally, but vertical orientation is also possible. The name of this testing apparatus originates from the shape of the heater. The heater is wound to a conical shape from an over 3 m long resistance heating wire packed in magnesium oxide refractory.
The flue gases from the burning sample are collected into the extraction hood situated directly above the heater. A flue gas fan is mounted in the flue gas line to set the flow
rate of combustion products. The gas sampling ring is situated before the fan in the flue gas line. Prior to reaching the gas analyzers, the gas sampled in the ring is first passed through two filters to remove particles, then through a cold trap and a drying agent to remove possible water. Between the gas sampling ring and the fan there is also a smoke measurement system. This measures the produced amount of smoke by a laser photometric beam.
The only analyzer needed to perform fundamental cone calorimeter experiments is the oxygen analyzer. Additional analyzers, such as carbon monoxide and carbon dioxide analyzers are usually fitted to gain better understanding of the burning process and to decrease the uncertainties in the results. For further analysis of combustion products and gas species for example a Fourier Transform Infrared spectrometer can be coupled through a heated line.